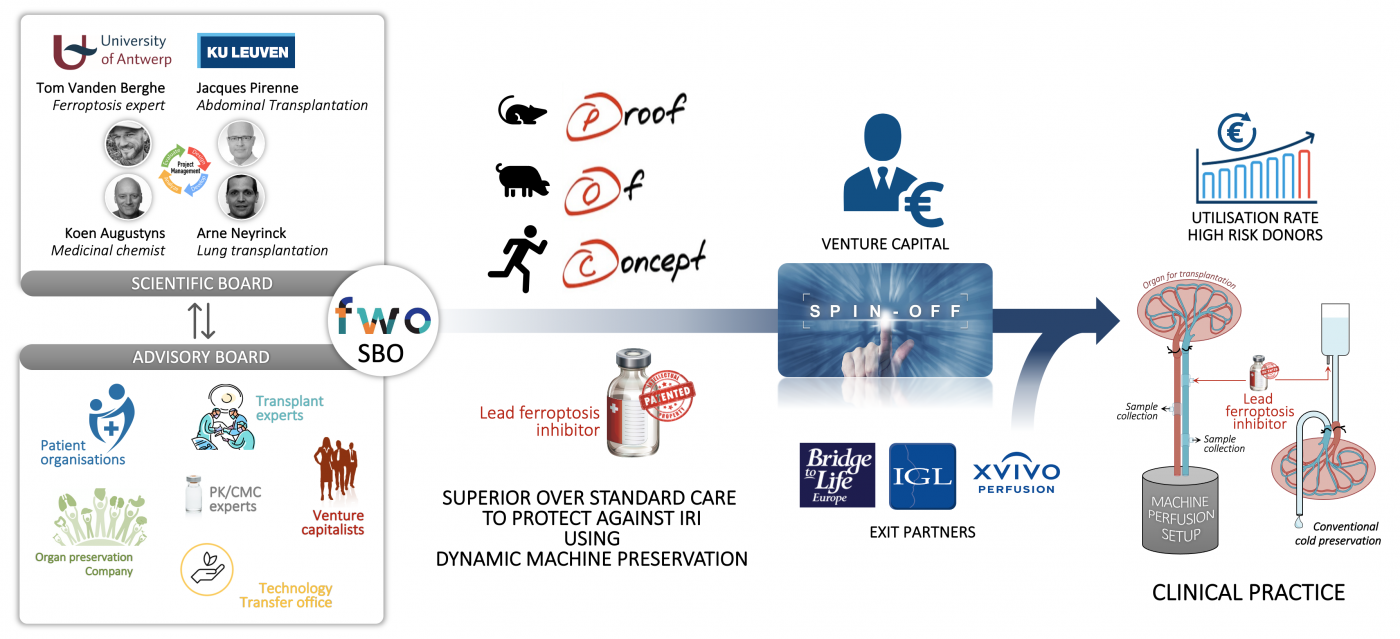
The success of transplantation is hampered by a shortage in suitable organ grafts and the adverse effects of ischemia reperfusion injury (IRI). Inflammation and cell death in the transplanted organ, caused by the activation of the innate immune system as part of the IRI process, leads to primary graft dysfunction (PGD). Transplant recipients that suffer from severe PGD have an increased risk for early and late morbidity and mortality.
The organ perfusion strategy was developed to increase the number of available grafts. During the ex situ phase between organ retrieval and transplantation, machine perfusion offers a unique window of opportunity for organ graft modulation to target IRI due to ferroptosis. Ferroptosis is an iron-dependent type of cell death in which oxidative stress initiates excessive lipid peroxidation of cellular membranes leading to cell death. Our in-house developed and patented third generation ferroptosis inhibitors show superior protection in preclinical models of organ injury and are therefore good drug candidates to block injury during transplantation.
In this project, we will firstly verify the efficacy of the lead ferroptosis inhibitor in protecting against ferroptosis using genetic organ injury models along experimental IRI or transplantation models in rodents. We will focus on liver, kidney and lungs as vital organs. Secondly, we will analyse the efficacy of adding our lead ferroptosis inhibitor to perfusate during normothermic machine perfusion preceding ex situ reperfusion in pigs. In parallel, we will evaluate the potential of ferroptosis inhibitors to recondition human organ grafts.
This research plan is a first step to implement ferroptosis inhibitory strategies in the clinical practice of transplantation, which is a steppingstone for building a spin-off case in ferroptosis therapeutics.